Stem Cell Technologies

- Professor
- KURISAKI Akira

- Assistant Professor
- TAKADA Hitomi

- INTO Atsushi

- Labs HP
- https://bsw3.naist.jp/kurisaki/
Outline of Research and Education
Pluripotent stem cells such as embryonic stem cells (ES cells) and induced pluripotent stem cells (iPS cells) are stem cells from the early stages of development that possess the ability for unlimited proliferation and differentiation into all cell types in the body. Due to their robust pluripotency and proliferative capacity, they are expected to be applied in regenerative medicine and drug discovery. However, because these stem cells are from the early stages of development, inducing differentiation into specific cells requires a long period, and sophisticated culture techniques are necessary for differentiation control. On the other hand, recent developments in culturing the stem cells in adult organs as organoids (mini-organs) and differentiating them into various cell types have enabled detailed studies of adult organ functions. Additionally, comparison between normal organoids with disease organoids enables to investigate the mechanisms of disease onset in detail within laboratory dishes. We particularly focus on analyzing the mechanisms of stem cell differentiation in organs, such as the stomach and lungs, and understanding of tissue development and regeneration.
Major Research Topics
Differentiation of Gastric Tissues
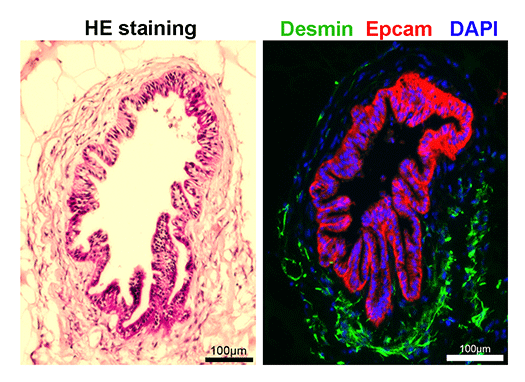
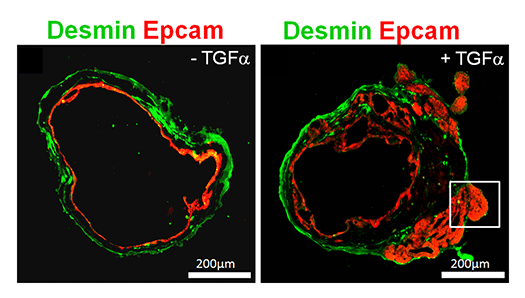
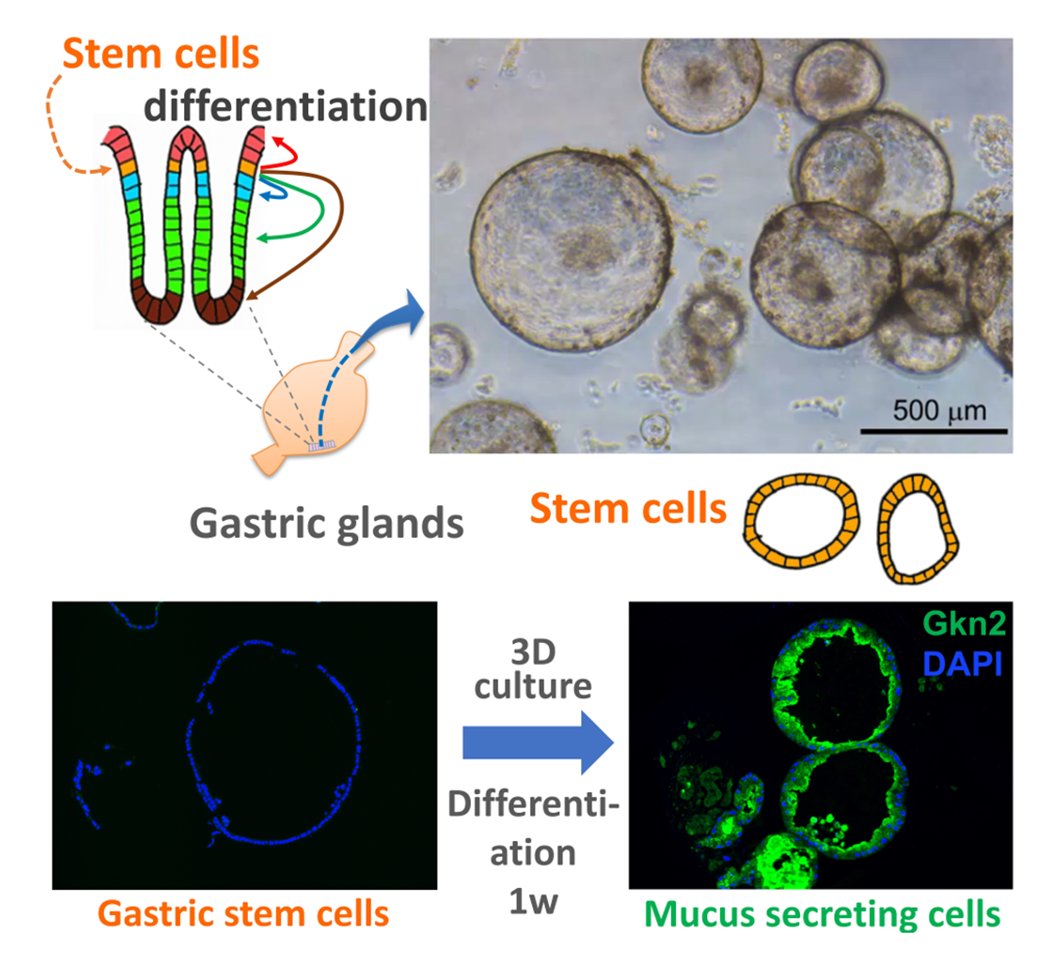
Although the stomach is a major digestive organ, the development mechanism is not well understood. It is believed that a single digestive tract is formed from the endoderm, the region of the stomach is determined, and further matures to form millions of glandular structures called gastric glands within the stomach lining. However, the mechanism behind this process remains largely unknown. We have reported methods for producing gastric tissue as a whole organoid from ES cells in vitro. Furthermore, we have recently developed methods to proliferate adult gastric tissue stem cells and differentiate them into functional cells. These methods serve as powerful tools for investigating the mechanisms behind gastric tissue formation. Additionally, since diseases such as gastric cancer lack good animal models, gastric tissues produced in vitro like this are also considered useful for studying the mechanisms of disease onset.
Differentiation of lung tissue and tissue regeneration
Like the stomach, the lung originates from the endoderm of the early embryo, where a lung bud emerges from the developing digestive tract and branches out to form the lung. While methods for differentiating lung tissue cells from human iPS cells have been studied, we are also advancing the development of methods to differentiate lung progenitor cells and bronchial ciliated epithelial cells, aiming to elucidate their differentiation control mechanisms.
Analysis of the Regulatory Mechanisms of the Undifferentiated State in Human iPS Cells
Human pluripotent stem cells are undifferentiated stem cells from the early stages of development that possess the ability to differentiate into various cell types. Using human iPS cells—a type of pluripotent stem cell—we are analyzing the regulatory mechanisms governing their undifferentiated state and cellular differentiation, specifically from the viewpoint of cell surface receptors.
References
- Intoh A. et al., Stem Cell Transl. Med. 13. 763-775, 2024
- Takada H. et al., Nature Comm., 14, 3750, 2023
- Hashimoto O et al., Cell Reports, 25, 1193-1203, 2018
- Sasagawa Y et al., Genome Biol. 19, 29, 2018
- Noguchi TK et al., Nature Cell Biology, 17, 984-993, 2015
- Watanabe-Susaki K et al., Stem Cells, 32, 3099-3111, 2014
- Seki Y et al., Proc. Natl. Acad. Sci. U S A, 107, 10926-10931, 2010

 NAIST Edge BIO
NAIST Edge BIO