Organelle Control

- Associate Professor
- KIMATA Yukio

- Assistant Professor
- KIMATA Yuki

- Labs HP
- https://bsw3.naist.jp/kimata/
Outline of Research and Education
Eukaryotes, such as plants, animals, and fungi, possess a wide variety of organelles, which vary in shape and activity depending on environmental conditions. Using yeast, a eukaryotic microorganism, we investigate the mechanisms by which the size and function of a predominant organelle, the endoplasmic reticulum (ER), are regulated.
Saccharomyces cerevisiae, a yeast species with a strong ethanol fermentation ability, is widely used in brewing, breadmaking, and bioethanol production. Because yeast is easily and rapidly subjected to various genetic and molecular biological experiments, it is frequently used as a model organism to elucidate biological phenomena that occur in other eukaryotic species. Our findings will be helpful for a deeper understanding of human diseases related to dysfunction of the ER, namely, ER stress. We also aim to establish industrially advantageous yeast strains carrying artificially enforced ER for improved production of ethanol, biopharmacectuals, and lipidic products.
Major Research Topics
Endoplasmic reticulum (ER) stress and unfolded protein response (UPR)
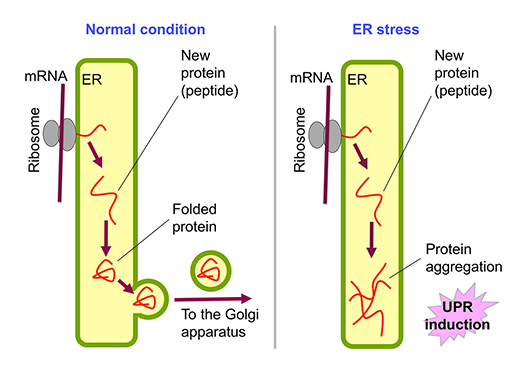
The ER is the cellular compartment in which secretory proteins are folded. Under non-stress conditions, they exit the ER and are transported to the cell surface via the Golgi apparatus (Fig.1, left). Biosynthesis of various lipidic molecules is also performed in the ER. On the other hand, ER stress causes accumulation of unfolded proteins in the ER (Fig. 1, right). To cope with such harmful conditions, eukaryotic cells provoke a cytoprotective response called the unfolded protein response (UPR).
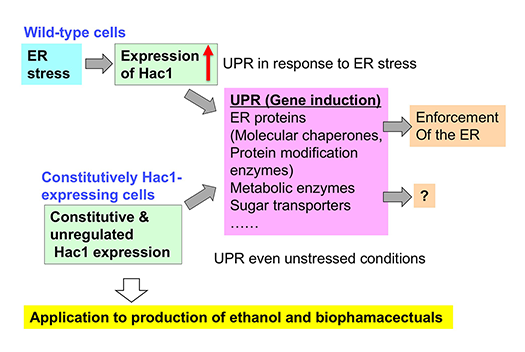
In yeast UPR, ER stress induces the nuclear transcription factor Hac1, which elevates the transcription levels of many genes, including those encoding ER-localized molecular chaperones and protein modification enzymes, leading to the enforcement of the ER (Fig.2: Wild-type cells). Hac1 also causes the induction of genes encoding metabolic enzymes and sugar transporters, the physiological consequences of which are obscure. Using omics technologies, we aim to elucidate what occurs in UPR-induced yeast cells to mitigate ER stress.
Yeast strains with the artificially enforced ER
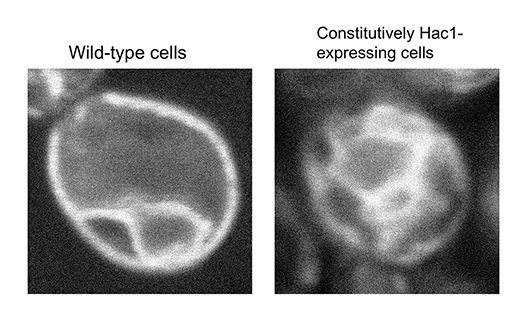
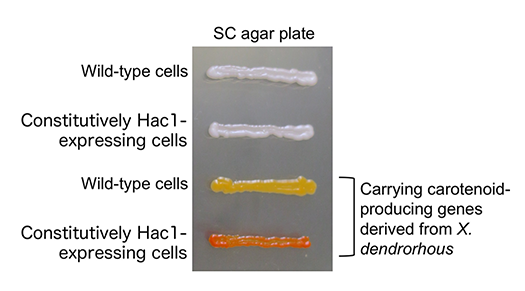
Human secretory proteins, such as insulin and cytokines, heterologously expressed in yeast cells are currently used as biopharmacectuals. Moreover, lipidic molecules, such as triglycerides and terpenoids, may be industrially produced from yeast in the near future. Since secretory proteins and lipids are produced in the ER, the yield of these commercially valuable biomolecules is anticipated to increase when yeast cells are modified to carry the ER with activated functions. To this end, we created mutant yeast strains that constitutively express Hac1 even under normal conditions (Fig. 2). The ER of the constitutively Hac1-expressing cells was highly expanded (Fig. 3). Moreover, the constitutively Hac1-expressing cells produced more secretory proteins and lipidic molecules, such as β-carotene, than wild-type cells (Fig. 4). Our current mission is to modify the constitutively Hac1-expressing cells toward high-yield production of commercially valuable bioproducts.
Ethanol fermentation and ER stress
Ethanol is known to act as a protein denaturant. We previously noted that, under our laboratory conditions, ethanol causes ER stress in yeast cells. Then, how does this affect industrial ethanol production, including brewing, by yeast? We will investigate the relationship between ethanol fermentation and UPR toward the high-yield production of ethanol.
ER stress induction by human pathogenic proteins in yeast cells
In some human neurodegenerative diseases, such as Parkinson's and Huntington’s diseases, unfolded or misfolded cytosolic proteins induce ER stress, leading to the aggravation of symptoms. We plan to express cytosolic pathogenic proteins in yeast cells and investigate the mechanism by which these proteins damage the ER.
Stress resistance of heat-tolerant yeast Kluyveromyces marxianus
ER stress and UPR can also be induced by culturing cells at high temperatures. K. marxianus can grow even at high temperatures of near 50 ℃. We investigate the molecular basis underlying the high heat resistance to K. marxianus. Moreover, our study on alcohol tolerance will contribute to the generation of K. marxianus strains suitable for biofuel production.

References
- Ishiwata-Kimata et al., Appl. Environ. Microbiol, 91, e0064425, 2025
- Geronimo et al., Front. Microbiol., 16, 1629132, 2025
- Fauzee et al., Front. Microbiol., 14, 1157146, 2023
- Ishiwata-Kimata and Kimata, J. Fungi (Basel), 9, 989, 2023
- Nguyen et al., Appl. Environ. Microbiol., 88, e01083-22, 2022
- Ishiwata-Kimata et al., Front. Cell Dev. Biol., 9, 743018, 2022