Plant Growth Regulation

- Professor
- UMEDA Masaaki

- Assistant Professor
- Zhang Ye

- KAWAMOTO Nozomi

- Labs HP
- https://bsw3.naist.jp/umeda/
Outline of Research and Education
Plants continuously form organs throughout their lives, a defining trait that distinguishes them from animals, which complete organ formation during early embryogenesis. Our research aims to elucidate the mechanisms by which plants—with their enduring and resilient vitality—maintain tissue and genome integrity, regulate cell division in response to environmental stress, and promote growth through DNA polyploidization and symbiotic interactions with microorganisms. Through these studies, we aim to uncover how plants sustain growth under fluctuating environments. This work will not only deepen our understanding of plant-specific survival and growth strategies but also contribute to the development of innovative technologies to increase food and biomass production.
Major Research Topics
Mechanisms for maintaining tissue and genome integrity
Plants can live for thousands of years and possess a remarkable capacity for organogenesis, continuously generating new organs throughout their lifespan. However, unlike animals, their cells do not develop cancer. To better understand these unique characteristics, we are investigating the mechanisms by which dividing cells maintain a high level of genomic stability. In addition, we are establishing experimental systems that induce cell death or overproliferation in specific cell types, with the aim of elucidating the regulatory mechanisms governing subsequent tissue regeneration and the suppression of cell division (Fig. 1). Through this research, we seek to uncover how plants—despite lacking brains or nervous systems—maintain the integrity of their organs, tissues, and genomes while flexibly adapting to their environment.
Mechanisms for suppressing cell division in response to environmental stress
Because plants are unable to move, they temporarily arrest cell division when exposed to environmental stress, redirecting energy toward stress adaptation. While this is a crucial survival strategy, it reduces productivity from an agricultural perspective and is therefore desirable to mitigate. We have recently identified a mechanism by which plants arrest the cell cycle at the G2 phase and halt growth in response to various environmental stresses (Fig. 2). By elucidating the signaling pathways that activate this checkpoint, we aim to develop crops capable of sustaining growth and maintaining productivity even under fluctuating environments.
Development of technologies to increase woody biomass production
Many plants undergo DNA polyploidization—a process in which DNA is repeatedly duplicated after cell division—thereby increasing cell and organ size. However, for reasons not yet understood, trees do not undergo DNA polyploidization. Therefore, we are working to develop technologies to enhance woody biomass production through tetraploidization. Currently, we are advancing research aimed at increasing trunk biomass by 30% using symbiotic microorganisms (Fig. 3). Furthermore, our recent findings indicate that changes in chromatin structure are critical for inducing DNA polyploidization, and we are working to elucidate the underlying molecular mechanisms. Through these studies, we hope to contribute to global-scale carbon recycling.
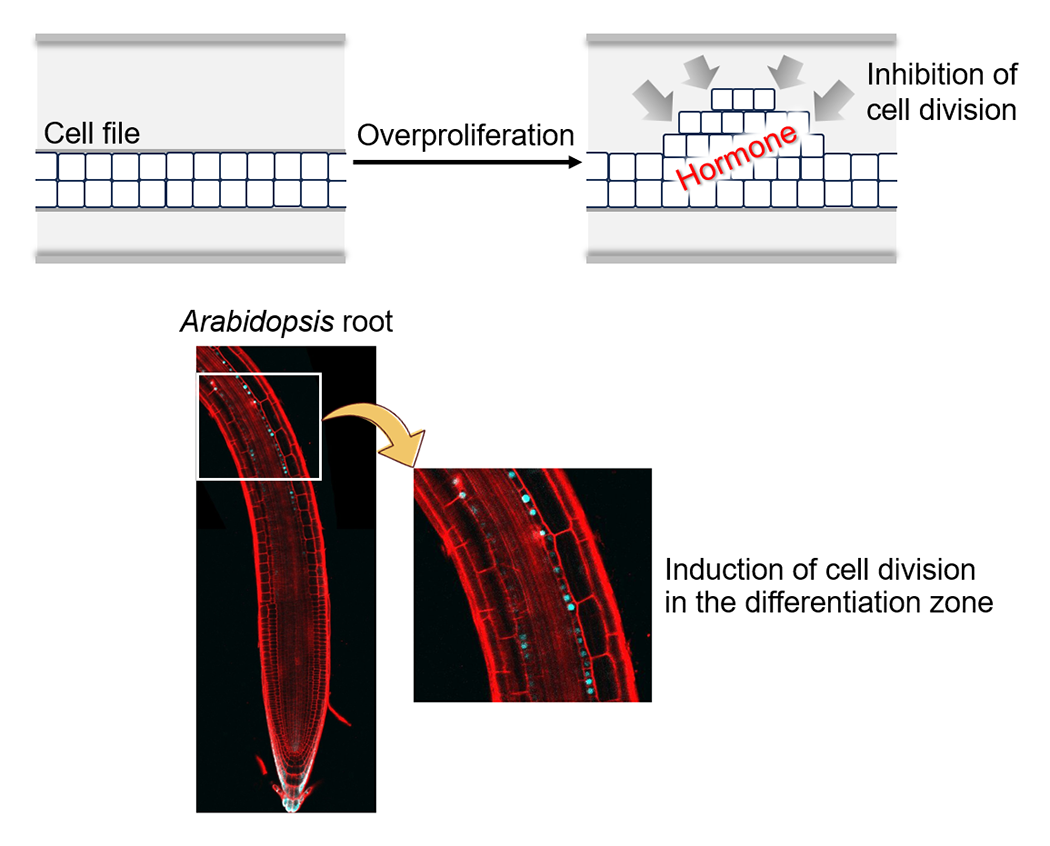
We have established an experimental system that induces cell division in a specific cell type within the differentiation zone of Arabidopsis roots. Using this system, we aim to elucidate the mechanisms by which signaling molecules, such as hormones, suppress cell division.
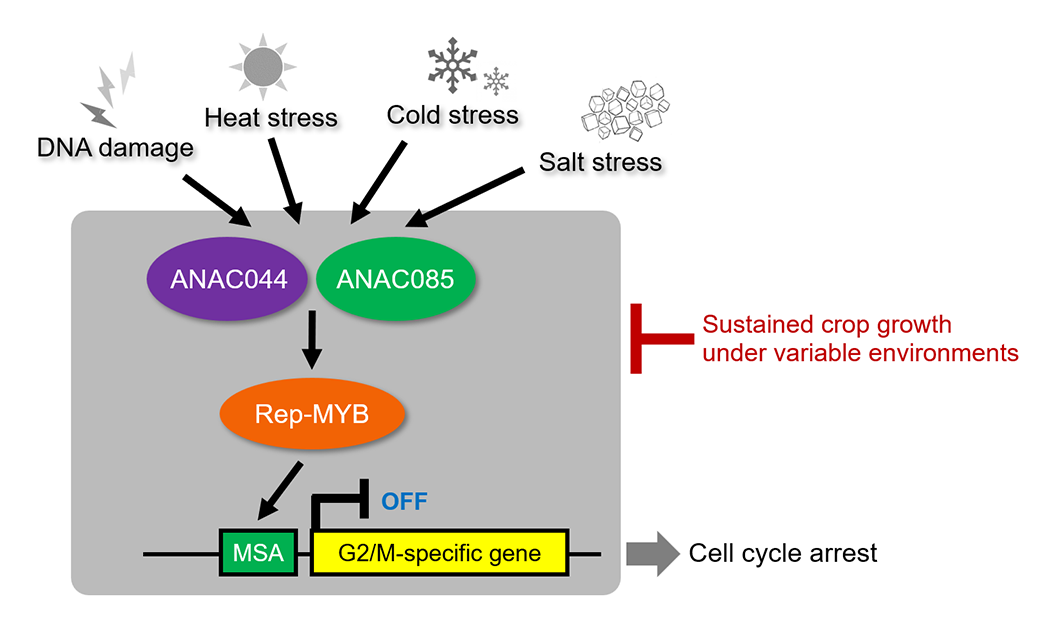
When exposed to environmental stresses such as DNA damage or high temperatures, plants activate a unique signaling pathway that arrests the cell cycle at the G2 phase. By modifying this regulatory system, we aim to develop crops capable of sustaining growth even under fluctuating environments.
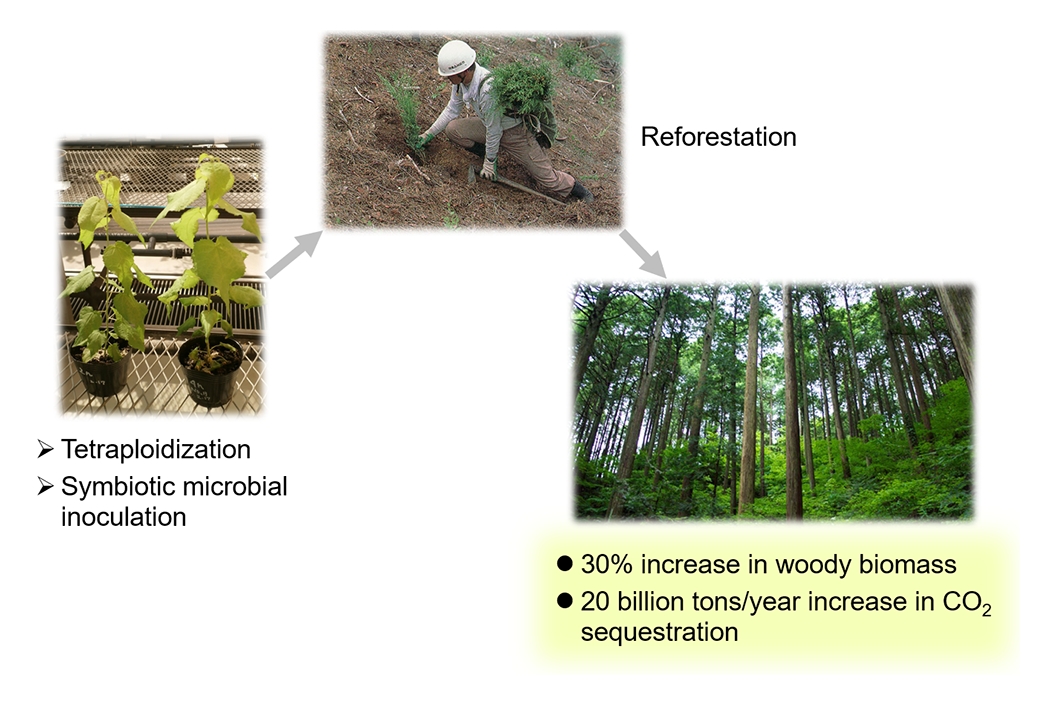
We are developing technologies to sustainably enhance trunk biomass production in plantations through the generation of tetraploid trees and the inoculation of symbiotic microorganisms. This approach could potentially offset more than half of the carbon dioxide emissions associated with global energy consumption.
References
- Takahashi N. et al., Plant Physiol., 198, kiaf303, 2025
- Echevarría C. et al., Nat. Plants, 11, 2145-2155, 2025
- Takahashi N. et al., J. Exp. Bot., 75, 1364-1375, 2024
- Takatsuka H. et al., J. Exp. Bot., 74, 3579-3594, 2023
- Takahashi N. et al., Sci. Adv., 7, eabg0993, 2021
- Shimotohno A. et al., Annu. Rev. Plant Biol., 72, 273-296, 2021
- Watanabe S. et al., Proc. Natl. Acad. Sci. USA, 117, 31500-31509, 2020
- Umeda M. et al., Curr. Opin. Plant Biol., 51, 1-6, 2019
- Takahashi N. et al., eLife, 8, e43944, 2019
- Takatsuka H. et al., Plant Physiol., 178, 1130-1141, 2018
- Ogita N. et al., Plant J., 94, 439-453, 2018
- Chen P. et al., Nat. Commun., 8, 635, 2017

 NAIST Edge BIO(
NAIST Edge BIO(