Research outcomes
Controlling cell differentiation on photo-designable synthetic collagen-like scaffolds
~A first step for creating “designer organs & tissues” ~
【Summary】
Regenerative medicine is one of the hot topics in scientific research. In rapidly aging societies throughout the world, demand for reconstructing the functions of impaired body parts has been moving into the limelight. There are two approaches for substituting impaired organs and tissues with new ones that have been a long-term dream in clinical applications.
- Creating organs and tissues by differentiation of stem cells.
- Making substitute organs and tissues using biocompatible materials.
Since the pioneering work on induced pluripotent stem (iPS) cells by Prof. Yamanaka (now at Kyoto University) and his students at NAIST, the former approach has gained high interest because iPS circumvents the ethical issues inherent in embryonic stem cells. However, although spatio-temporally controlled differentiation of stem cells in vivo has been eagerly pursued, it has been difficult.
Stem cells have an ability to differentiate into many kind of cell types, and the differentiation is highly affected by external factors including peptides, proteins and low molecular compounds. If those factors could be administered in a spatiotemporally controlled manner, the differentiation of stem cells could be controlled. By dictating the cell differentiation of several cell types, the replacement of certain types of organs or tissues may be possible.
Scaffold/extracellular matrix is another important factor for organ and tissue regeneration. One of the major components in extracellular matrices is collagen. At NAIST, Prof. Tanihara’s group has developed a chemically synthesized collagen-like polypeptide, poly(Pro-Hyp-Gly) which has a similar structure with type I collagen. The polypeptide has been used for cosmetics and other healthcare applications.
This year, Dr. Mime Kobayashi and co-workers have developed biocompatible scaffolds that enable cell fate control with visible light. The scaffolds are based on the synthetic collagen-like polypeptide. Bioactive peptides, which can be dissociated by laser irradiation, were conjugated to the scaffolds. This strategy enables optical control of the attachment and detachment of functional peptides in pre-designed shapes. Using bone forming peptide (BFP) and rat bone stromal cells, the initial stage of bone formation was confirmed. Using other signaling molecules and three-dimensional collagen-like scaffolds, controlled differentiation of stem cells into specific organs or tissues can be achieved.
Towards application of the scaffold, collaborations with overseas researchers are already underway. A research proposal with Dr. Retno Wahyu Nurhayati from Universitas Indonesia led to her recognition as a recipient of L’ORÉAL – UNESCO for Women in Science National Fellowship Awards 2017.
These accomplishments are the results of a central focus on interdisciplinary research at NAIST. This work is a first step toward the utilization of biocompatible materials as platforms to direct stem cell differentiation.
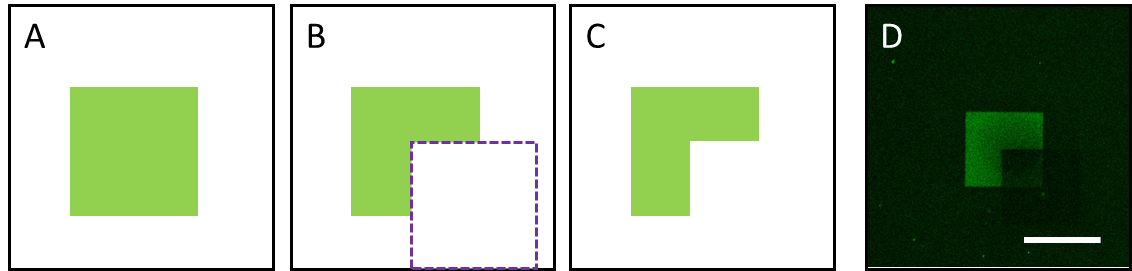
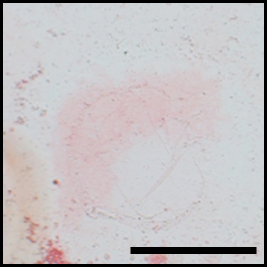
Comment by Dr. Kiichiro Tomoda, a founding member of Yamanaka lab at the Gladstone Institutes
“If spatio-temporal control of iPS cell differentiation in vivo is achieved, it will be possible to create organs or tissues at will. This result is a great first step towards the goal.”
【Resource】
Title: Optical control of cell differentiation on synthetic collagen-like scaffolds
Authors: Mime Kobayashi*, Ryosuke Honda, Tsuyoshi Ando, Masao Tanihara
Publication: Journal of Biomaterials Science, Polymer Edition (2019) 30:1161-1171.
DOI:10.1080/09205063.2019.1622845
URL: https://www.tandfonline.com/doi/full/10.1080/09205063.2019.1622845
【Patent】
Substrate for stem cell culture, and culture method using same
Inventors: TANIHARA, Masao; SATO, Narutoku; KOTOKU, Tomomi; SHIBASAKI, Yoshiaki
International publication No.: WO/2013/002311 January 3rd, 2013
【Funding】
Takeda Science Foundation
The Green Photonics Project and the Global Collaboration Program in Nara Institute of Science and Technology sponsored by the MEXT, Japan.
【Plant Stem Cell Regulation and Floral Patterning Lab】
https://bsw3.naist.jp/eng/courses/courses112.html
Lab website: https://bsw3.naist.jp/ito/?cate=446
( December 20, 2019 )
