Research outcomes
Novel protein transport mechanism for axon outgrowth
Nerve cells extend long processes called axons and form complicated networks in the brain. For the extension and maintenance of axons, neurons have to deliver proteins from their cell body to the tip of axon. It is well known that the motor proteins kinesins, dyneins and myosins, which walk along cytoskeletons, play key roles in the transport of vesicle-anchored proteins and some cytoplasmic proteins. In contrast, the molecular basis for the transport of actin and actin-associated proteins along axons remains poorly understood.
The research group of Prof. Naoyuki Inagaki, Hiroko Kastuno in the Laboratory of Systems Neurobiology and Medicine, Graduate School of Biological Sciences, Associate Prof. Yoichiroh Hosokawa in Graduate School of Materials Science, Prof. Kazushi Ikeda in Graduate School of Information Science in NAIST, Associate Prof. Yuichi Sakumura in Aichi Prefectural University, and Prof. Kensaku Mizuno in Tohoku University found a protein transport mechanism for actin and actin-associated proteins along axon.
The group analyzed actin waves, which translocate actin and actin-associated proteins along neuronal axons toward the axonal tip (Fig. 1). They found that arrays of actin filaments constituting waves undergo directional assembly and disassembly, with their polymerizing ends oriented toward the axonal tip, and that the lateral side of the filaments is mechanically anchored to the adhesive substrate. A combination of live-cell imaging, molecular manipulation, force measurement and mathematical modeling revealed that the actin migration is driven by directional assembly and disassembly of actin filaments and their anchorage to the substrate. Actin-associated proteins co-migrate with actin filaments by interacting with them. Furthermore, blocking this migration, by creating an adhesion-free gap along the axon, disrupts axonal protrusion.
Their findings identify a novel molecular mechanism that translocates actin and associated proteins toward the cell’s leading edge, thereby promoting directional cell motility. The findings also provide a new insight for axon formation and outgrowth.
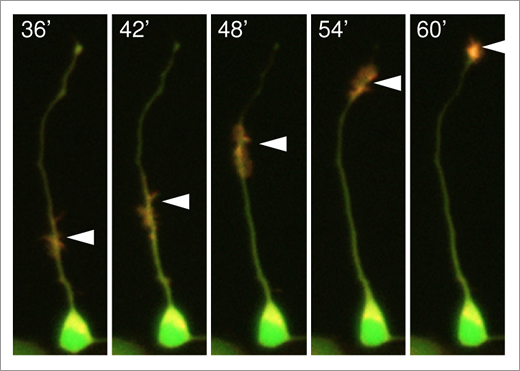
Fig.1 Actin waves transported along an axon (arrowheads).
Reference
Katsuno, H., Toriyama, M., Hosokawa, Y., Mizuno, K., Ikeda, K., Sakumura, Y, and Inagaki, N. (2015)
Actin migration driven by directional assembly and disassembly of membrane-anchored actin filaments, Cell Reports 12, 648-660.
[ Press Release ] July 17, 2015
( January 18, 2016 )
